Municipal Tap Water in the Aquarium
Most people use tap water in their tanks; it is cheap and easy to use. Unfortunately (for aquarists), local water companies add chemicals to the water to make it safe to drink (e.g., chlorine or chloramine to kill bacteria). More recently, concern about water flowing through older lead pipes has caused some water utilities to add pH-raising chemicals to the water (because lead dissolves less readily in alkaline water). Consequently, tap water must be specially treated before it can safely be used in fish tanks.
Another potential problem concerns variability in the chemical properties of your water supply over time (e.g., month-to-month). Some water districts don’t have enough water themselves, forcing them to purchase additional water from neighboring water districts in times of shortages. If this water has a different chemical properties (e.g., hardness), your tap water’s chemistry will vary as well. As a common example, high bacteria levels are more of a problem in summer than winter, especially in warmer climates. Consequently, it is not uncommon for water companies to use more chlorine in summer months to keep bacteria in check. Even such factors as local weather can have an impact; heavy rains may cause the hardness of your water supply to decrease as local reservoirs fill.
In general, chlorine and chloramine are the two additives that cause the most problems. Note that these two substances are VERY DIFFERENT! Be sure you know what is in your tap water and treat appropriately.
Chlorine
In the US, EPA guidelines require that tap water at any faucet contain a minimal chlorine concentration of 0.2 ppm, and stringently limits the concentration of bacteria (which may require more than 0.2 ppm chlorine to keep in check). Because chlorine breaks down over time, the chlorine concentration of the water that comes out of your tap will be lower than that put in at water plant. Thus, the exact concentration at your faucet depends on how far you are from the water plant, how long it takes the water to travel from the water plant to your house, how much chlorine is initially added, etc.
Chlorine at high concentrations is toxic to fish; at lower concentrations, it stresses fish by damaging their gills. Concentrations of as little as 0.2-0.3 ppm kill most fish fairly rapidly. To prevent stress, concentrations as low as 0.003 ppm may be required. Fortunately, chlorine can easily be removed from water by the chemical sodium thiosulfate, readily available at fish stores under various brands. Sodium thiosulfate neutralizes chlorine instantly. Note that there are many “water treatment” products that are advertised as “making tap water safe”. Read labels carefully. Inevitably, the ones that neutralize chlorine all contain sodium thiosulfate, plus other substances that may or may not be useful. If your water only contains chlorine (as opposed to chloramine), sodium thiosulfate is all you need. The most cost-effective treatments use only 1 drop per gallon of water. Most other water treatments are much more expensive in the long-term; they may require a teaspoon of treatment (or more) per gallon!
Chlorine is relatively unstable in water, escaping to the atmosphere on its own. Water left in a bucket (or tank) with adequate water circulation (e.g. filter or airstone) will be free of chlorine in 24 hours or less.
Many netters report that they perform partial water changes without ever treating their tap water to remove chlorine. Keep in mind that even though fish show no APPARENT ill effects from untreated water, that doesn’t mean that the chlorine isn’t stressing your fish. How much stress depends on how much chlorine is introduced to the tank, which depends on many factors (including the percentage of new water added). Because chlorine removers are so cheap (pennies per usage), the insurance they provide should not be passed up.
Chloramine
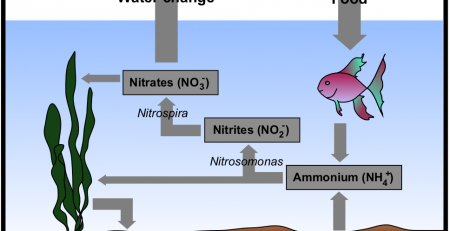
One problem with using chlorine to treat water is that it breaks down relatively quickly. Another concern with the use of chlorine is that it can combine with certain organics (that may or may not be present in your water) forming trihalomethanes, a family of carcinogens. Consequently, many water companies have switched from using chlorine to using chloramine. Chloramine, a compound containing both chlorine and ammonia, is much more stable than chlorine.
Chloramine poses two significant headaches for aquarists. First, chlorine-neutralizing chemicals such as sodium thiosulfate only neutralize the chlorine portion of the chloramine, neglecting an even bigger problem: deadly ammonia. The consequences can be devastating to fish. Although a tank’s biological filter will (eventually) convert the ammonia to nitrate, the time it takes to do so may be longer than what your fish can tolerate.
The second problem relates to water changes. One of the primary reasons for doing regular water changes is to remove nitrates that build up. If your replacement tap water contains ammonia, you’ll be putting nitrogen right back into your tank and it will be impossible to reduce the nitrates below the concentration in your tap water. Fortunately, tap water concentrations are relatively low (1 or 2 ppm); you are more likely to have a much higher concentration of nitrate in your tank.
Chloramine can be safely neutralized through such products as Amquel, which neutralize both the ammonia and chlorine portions of the chloramine molecules. The neutralized ammonia will still be converted to nitrates via a biological filter.
Another method for neutralizing chloramine is to age the water while simultaneously performing biological filtration. For example, get an appropriately-sized (plastic) garbage can, fill it with tap water, dechlorinate it with sodium thiosulfate, and then connect an established biological filter to it. Just as in your tank, the bio filter will convert the ammonia to nitrate, after which it can safely be added to your tank. Note: you must add sodium thiosulfate to neutralize the chlorine; otherwise, the chloramine will kill the bacteria in your biological filter.
Alternatively, the ammonia can removed by filtering the water through zeolite or carbon before adding it to your tank. [Note: folks report mixed success with this. If you have concrete (positive or negative) experience to report, please notify the FAQ maintainers.]
Other water impurities you should be aware of
In addition to the additives described above (chlorine and chloramine), municipal water may (or may not!) contain other elements that the aquarist may need to know about. Water in some locations actually contains nitrates. In some places, water contains elevated concentrations of phosphates (1 ppm or more). High phosphate has been linked to algae problems, and a comprehensive algae control strategy may require removing phosphates. High levels of iron (1 ppm or more) have also been linked to thread algae. Consult the algae section of this FAQ for more details.
How to Find out What Your Local Water Company Adds to Your Tapwater
The quick answer is to ask someone who knows. A local fish store (if they reside in the same water district as you do) should be able to tell you. Alternatively, call your local water utility. Ask to speak with the “water chemist”. Tell them you are an aquarist and want to know about the pH, GH, and KH of your water, as well as how much the water characteristics vary from month to month. Finally, (in the US) if you really want details, have them send you a copy of the periodic water report they are required to generate for the EPA. It contains a detailed listing of exactly what your water contains and in what concentrations (e.g., iron, nitrates, phosphates, etc.). By law, the report is available for public inspection.
Well Water
You may have access to well water instead of municipal tap water. One advantage with well water is that you don’t need to deal with chlorine and chloramine. On the other hand, well water is frequently (much!) harder than water available through local utilities. In addition, the only way to know its composition (GH, KH, etc.) is to run tests on it yourself. Alternatively, there are companies to which you can send water samples that will perform a detailed analysis of its contents (for $20-100).
One potential problem with using well water is that it frequently contains high concentrations of dissolved gases (which may be dangerous to fish). For example, well water is frequently supersaturated with CO2, which lowers the water’s pH. Once the CO2 escapes, the pH will increase. Fish shouldn’t be subjected to this temporary pH fluctuation. For safety, aerate well water thoroughly for several hours before adding it to your tank.










Leave a Reply
You must be logged in to post a comment.